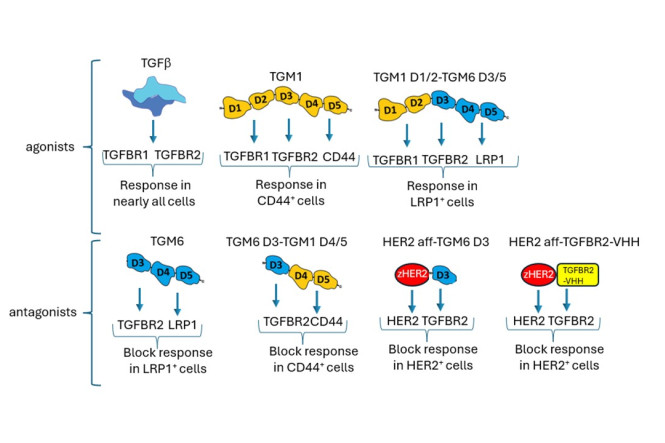
TGM6 binds to TGFBR2 (but not TGFBR1) and to LRP1 via separate domains. When TGM6 binds LRP1, it directs TGFBR2 to lysosomal degradation. The modular architecture of TGMs enabled us to create TGM1/6 chimeras with distinct cell-specificities and functions. Additionally, we engineered a TGFBR2 nanobody (VHH) that, when fused to an antibody targeting a specific co-receptor, selectively blocks TGFβ signaling in human cells. This approach may enable precise targeting of dysregulated TGFβ in cancer and fibrosis, potentially minimizing the severe side effects associated with conventional, broad-acting TGFβ inhibitors. We are excited about the potential of this modular designer agent strategy to advance targeted therapies
Read the full story on how we are leveraging parasite evolution to revolutionize drug development!
See our paper in Advanced Science https://lnkd.in/eypXYUJv